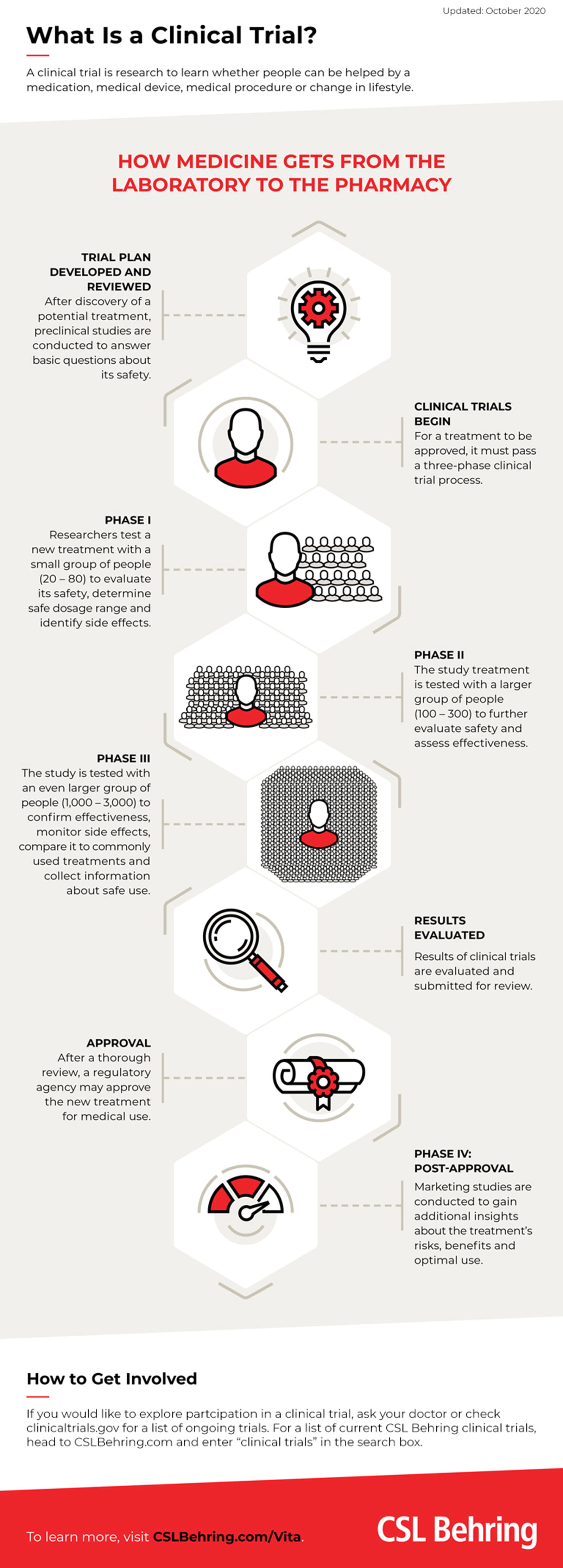
In the United States, clinical trials – the rigorous process for testing new treatments – include four phases, but Phase 3 is particularly important.
In Phase 3, the study drug or treatment is given to large groups of people and, according to the U.S. National Institutes of Health (NIH), the potential treatment is tested to:
- confirm its effectiveness
- monitor side effects
- compare it to commonly used treatments
- collect information that will allow the drug or treatment to be used safely
Last week, the CoVIg-19 Plasma Alliance, along with the U.S. National Institute of Allergy and Infectious Diseases (NIAID) at the National Institutes of Health (NIH) announced the start of a Phase 3 clinical trial to evaluate the safety and efficacy of an investigational anti-SARS-CoV-2 polyclonal hyperimmune globulin (H-Ig) medicine. The hyperimmune could potentially treat patients who have serious complications from COVID-19.
Learn more about the hyperimmune clinical trial.
The potential treatment, derived from plasma donated by patients who recovered from COVID-19, contains antibodies believed to be useful in fighting novel coronavirus infections. The potential hyperimmune, unlike a direct plasma transfusion, is intended to be a shelf-stable medicine that could be kept on hand at hospitals.
This summer, clinical doses of the potential treatment were manufactured in CSL Behring’s Bern, Switzerland, facility and sent to another CSL Behring site in Marburg, Germany, for packaging. In July, production began in Australia at the company’s Broadmeadows facility on a COVID-19 Immunoglobulin-VF, a hyperimmune treatment that has the potential to treat hospitalized Australian patients with COVID-19, particularly those whose illness is progressing toward the need for ventilation.
A group of leading plasma companies, with founding members CSL Behring and Takeda, formed the CoVIg-19 Plasma Alliance earlier this year.